 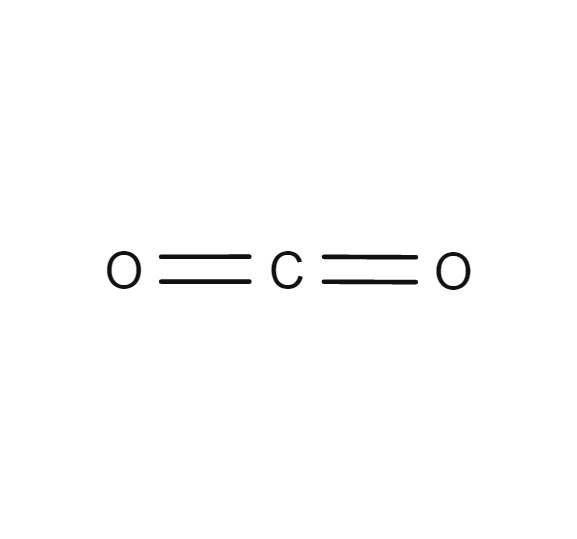
Other human activities are biomass burning and cement production. Without them, carbon dioxide would not be utilized at most and thereby accumulate in the atmosphere. Trees and other photosynthetic organisms serve as the major sink of carbon dioxide. This is largely due to the combustion of fossil fuels. At the onset of the Age of Industrialization, the concentration of carbon dioxide in the atmosphere increased to about 43%. One of the major factors that led to this rise is human activities. The concentration of carbon dioxide in the atmosphere has risen through the years. Thus, carbon dioxide is considered an important greenhouse gas, more than methane. Although carbon dioxide is just next to methane in terms of producing a greenhouse effect per volume, it is longer-lived than methane and occurs in a much higher concentration. These two are the major factors responsible for the greenhouse effect. In the atmosphere, carbon exists mainly in the form of carbon dioxide and methane. Carbon cycles through the Earth’s lithosphere, hydrosphere, and atmosphere. The carbon cycle is a biogeochemical cycle depicting carbon exchanges on Earth. It is a nonflammable gas.Ĭarbon is the fourth most abundant element in the Universe. At standard temperature and pressure, its density is 1.98 kg/m 3. at pressures below 5.1 atm.Ĭarbon dioxide has a molar mass of 44.01 g The liquid carbon dioxide returns into its gaseous state when the pressure drops, i.e. Thus, carbon dioxide is present not just in the Earth’s atmosphere but also in oceans, seas, rivers, lakes, groundwater, glaciers, and ice caps. The chemical reaction is as follows: CO 2 + H 2O ⇌ H 2CO 3. When it reacts to water, it forms carbonic acid, which is a weak acid. 2Ĭarbon dioxide is soluble in water, however, only when pressure is maintained. Thus, no net shifting of electrons occurs in any direction and as such there is no net charge build up on any of the atoms. The two oxygen atoms have equal force or electronegativity as they pull the electron density from the carbon at 180°. There is no unequal sharing of valence electrons. However, it does not have C-C and C-H covalent bonds present in organic compounds.ĭue to the linearity and centrosymmetry of the carbon dioxide, this compound is nonpolar. Carbon dioxide has a carbon atom and can be produced by a living thing through respiration. Nevertheless, carbon dioxide is an exception to this generalized rule. It sublimes at -78.5☌ at normal atmospheric pressure.Ī general depiction of an inorganic compound is one that lacks carbon atoms and archaically, not produced by a living thing ( Vitalism). Apart from gas, carbon dioxide may also occur in liquid and solid states. Its chemical formula is CO 2.Ĭarbon dioxide is a colorless, odorless, incombustible gas. The configuration of the compound is linear and centrosymmetric. 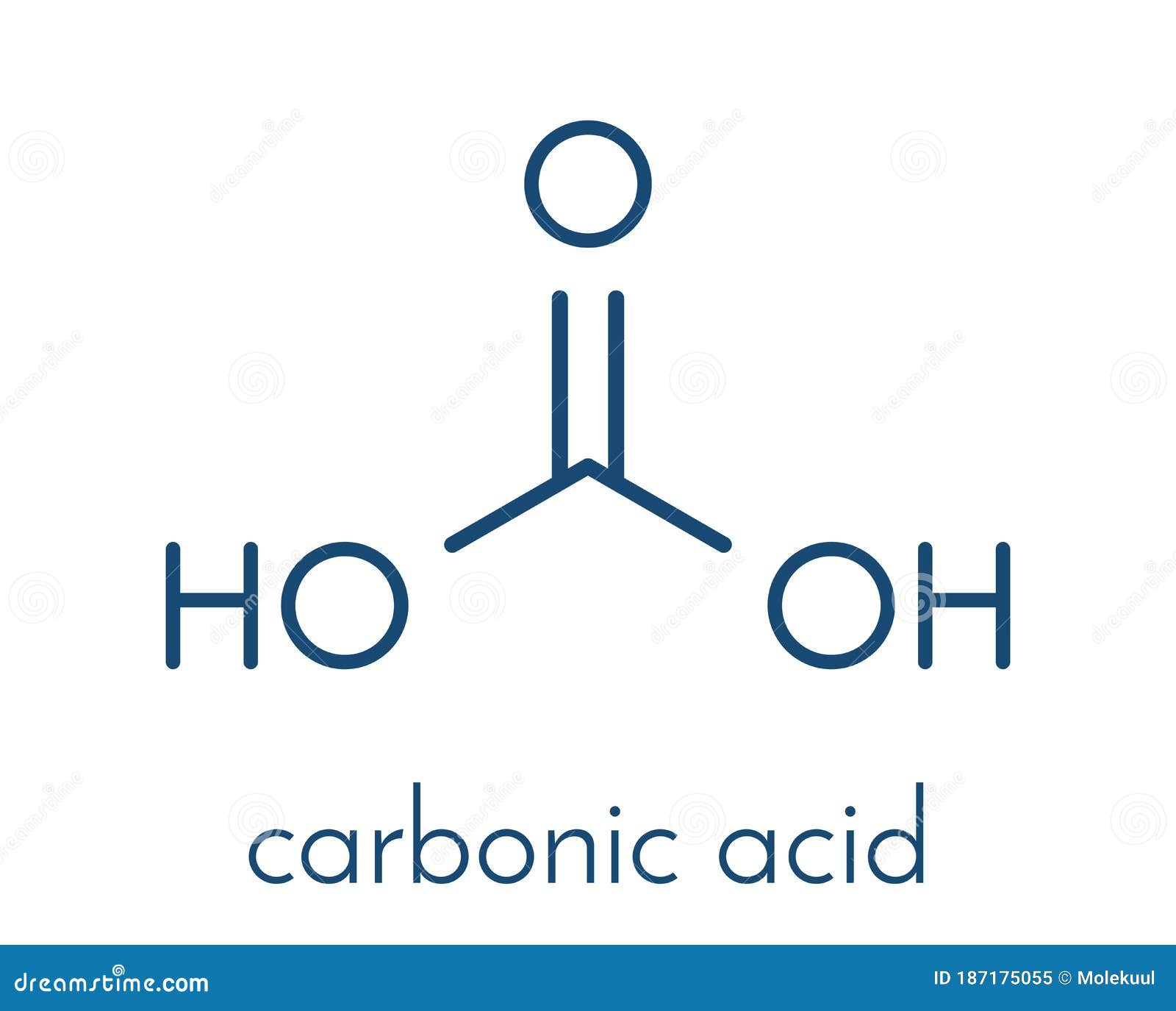
Each of the oxygen atoms is attached to the central carbon atom by a double covalent bond. Now, it is called “carbon dioxide”.Ĭarbon dioxide is a chemical compound made up of a carbon atom and two oxygen atoms. In 1803, John Dalton (English chemist) proposed that this substance is comprised of one carbon atom and two oxygen atoms. He also identified it from exhaled breath and described it as denser than the air faintly acidic. 1 In 1756, Joseph Black, a Scottish physician, noticed this gas when heating calcium carbonate (CaCO 3). He thought that it was an element or a single substance. He, them, called the vapor “wood gas” ( spiritus sylvestris). In 1630 or so, Jan Baptist van Helmont, a Flemish chemist, identified a vapor distinct from the air when he burned charcoal in a closed vessel. Carbon dioxide (CO 2) became the first gas that was distinguished from ordinary air. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
